Tuesday, 12 May 2026
Breakthrough study uncovers how photocatalytic and photothermal processes work together, setting the stage for next-generation carbon recycling technologies
As global carbon dioxide (CO₂) emissions continue to rise, scientists are intensifying efforts to develop technologies that can transform greenhouse gases into useful fuels. Researchers at Chiba University have now reported a significant advance by uncovering how light-driven and heat-driven processes interact to dramatically improve the conversion of CO₂ into methane (CH₄), a key step toward sustainable energy systems.
In a study published in the Journal of the American Chemical Society, a team led by Yasuo Izumi achieved one of the highest methane production rates reported to date using a Ru–Ni–ZrO₂ catalyst system. Beyond performance, the research addresses a long-standing challenge in the field by clarifying whether CO₂ reduction is driven primarily by true photocatalytic effects—where light excites electrons—or by photothermal effects, where light generates heat that accelerates reactions.
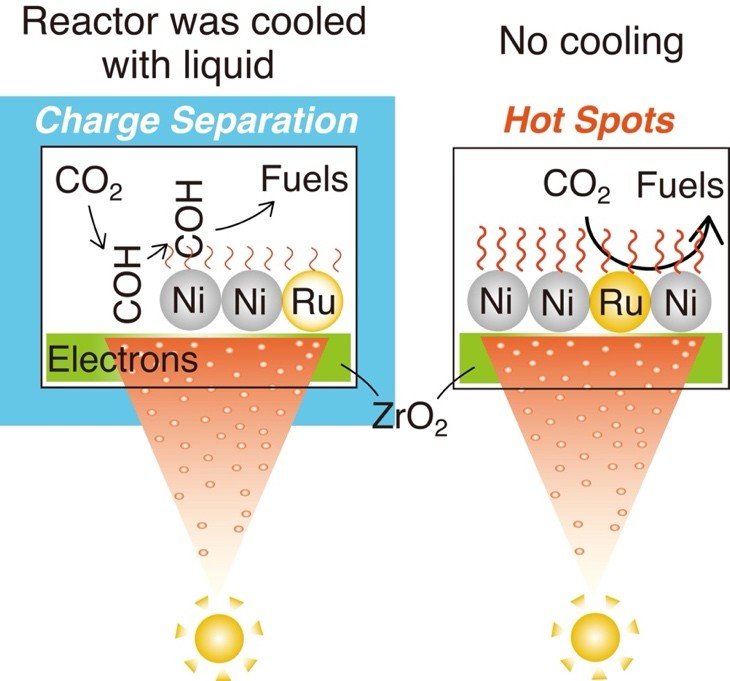
To resolve this, the researchers designed controlled experiments in which catalysts were exposed to ultraviolet–visible light under carefully regulated temperature conditions. By maintaining the system at low temperature in some cases and allowing it to heat naturally in others, they were able to distinguish the contributions of each mechanism and observe how they interact under different conditions.
Their findings show that CO₂-to-methane conversion proceeds through two distinct yet interconnected pathways. Under cooler conditions, the reaction is dominated by photocatalysis, where light excites electrons on the zirconia surface, creating reactive intermediates that are subsequently converted into methane through hydrogenation steps at nickel sites. In contrast, under higher temperature conditions without cooling, photothermal effects become dominant, enabling CO₂ molecules to be directly activated and dissociated on ruthenium–nickel active sites with significantly lower energy barriers.
Crucially, the study reveals that these two processes do not operate in isolation but instead reinforce each other. Under strong light irradiation, localized hotspots form on the catalyst surface, where elevated temperatures and active charge separation coexist. At these sites, methane production rates exceed what would be expected from purely thermal reactions, demonstrating a synergistic effect that enhances overall efficiency.
Using this combined mechanism, the Ru–Ni–ZrO₂ catalyst achieved methane production rates of up to 10 millimoles per gram per hour, placing it among the most efficient systems developed so far. The results also show that the balance between photocatalytic and photothermal contributions can be tuned by adjusting light intensity and temperature, offering a practical pathway for optimizing performance.
With global CO₂ emissions reaching record levels, technologies that can convert carbon dioxide into usable fuels are increasingly important for enabling a circular carbon economy. Methane produced through such processes can serve as an energy carrier while simultaneously reducing atmospheric carbon levels, making this approach particularly attractive for sustainable energy strategies.
Looking ahead, the research team aims to extend their methodology to produce more complex and valuable chemicals, including multi-carbon compounds and alcohols, using sunlight-driven catalytic systems. By providing a clear understanding of how light and heat work together in CO₂ conversion, this study offers a foundation for designing next-generation catalysts that are both more efficient and more scalable.
This work marks a critical step forward in bridging the gap between laboratory research and practical carbon recycling technologies, highlighting the potential of integrated photocatalytic and photothermal systems in addressing global climate and energy challenges.